Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Inoue, Akihiko
Nanotech 2010, Vol.3, p.576 - 578, 2010/00
The dissolution of plutonium dioxide (PuO ) is of a fundamental importance from the point of view of energy as well as enviroment. In this work the mechanism of mediated electrochemical dissolution of semiconducting plutonium dioxide (PuO
) is of a fundamental importance from the point of view of energy as well as enviroment. In this work the mechanism of mediated electrochemical dissolution of semiconducting plutonium dioxide (PuO ) particles in nitric acid (HNO
) particles in nitric acid (HNO ) was studied on the basis of experimental kinetics published so far. It was found that the rate-controlling reaction is an interfacial electron transfer process between the solid tetravalent plutonium Pu
) was studied on the basis of experimental kinetics published so far. It was found that the rate-controlling reaction is an interfacial electron transfer process between the solid tetravalent plutonium Pu (s) and an aqueous metal ion such as reductive U
(s) and an aqueous metal ion such as reductive U (aq) or oxidative Ce
(aq) or oxidative Ce (aq) ion. The rupture of covalent bond of Pu-O of the possible [PuO
(aq) ion. The rupture of covalent bond of Pu-O of the possible [PuO ]
]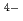 cluster in the (110) plane of the cubic PuO
cluster in the (110) plane of the cubic PuO lattice by electron transfer is decisive for the dissolution process. The role of HOMO and LUMO of the [PuO
lattice by electron transfer is decisive for the dissolution process. The role of HOMO and LUMO of the [PuO ]
]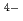 cluster is discussed with regards to the oxidative and reductive dissolution respectively.
cluster is discussed with regards to the oxidative and reductive dissolution respectively.